|
Product Name |
CAS No. |
|
Fumaryl diketopiperazine |
176738-91-3 |
Product Name (品名): | Fumaryl diketopiperazine 口服胰岛素载体 || 富马酰二酮哌嗪 |
| Appearance | White to off-white powder |
Molecular Formula (分子式): | C20H28N4O8 |
Molecular Weight (分子量): | 452.46 |
| Purity (纯度): | 98% |
| Storage | store at 2-8°C |
Contact Telephone (联系电话): | 023-677770219 |
Contact | Sales Department |
| Contact Email: | sales@chemieliva.com |
| Structure | |
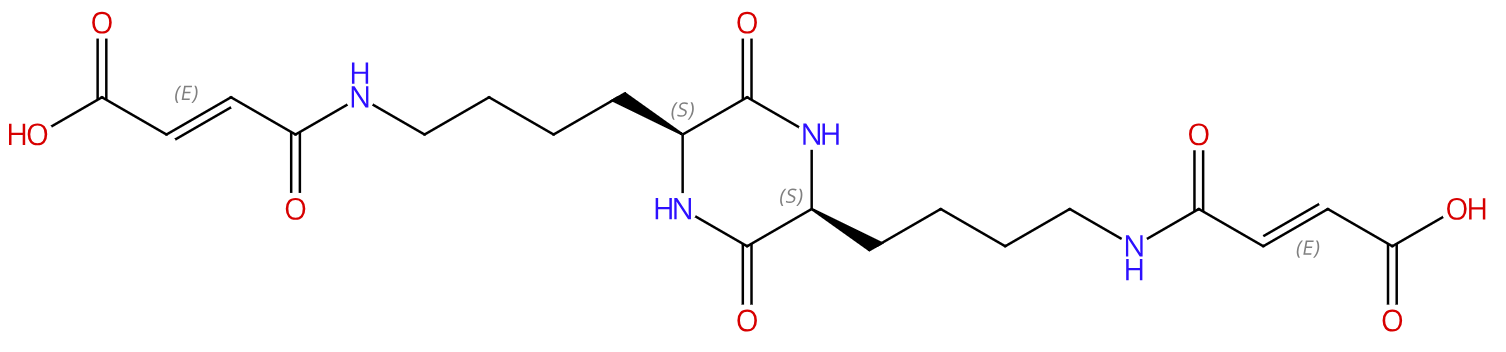 | |
Main Function
| |
Key Words:
176738-91-3 ||Chemieliva Pharmaceutical Co., Ltd || 重庆福腾医药|| 176738-91-3 seller || 176738-91-3 supplier || 176738-91-3 manufacturer || 176738-91-3 producer || 176738-91-3 price || 176738-91-3 China || 176738-91-3 India || 176738-91-3 MSDS || 176738-91-3 COA || CAS 176738-91-3 || buy 176738-91-3 || 176738-91-3 offer || 176738-91-3 seller || sell 176738-91-3 || selling 176738-91-3 || 176738-91-3 factory || 176738-91-3 factory || 176738-91-3 data || China 176738-91-3 || 176738-91-3 export || 176738-91-3 offer || 176738-91-3 China supplier || 176738-91-3 China Factory || 176738-91-3 Chinese supplier || 176738-91-3 Chinese factory || 176738-91-3 Chinese manufactuer || 176738-91-3 offer || 176738-91-3 quotation || 176738-91-3 production capacity || 176738-91-3 CPHI || offer 176738-91-3 || 176738-91-3 supplying || 176738-91-3 SDS || 176738-91-3 COA || 176738-91-3 packing || 176738-91-3 picture








